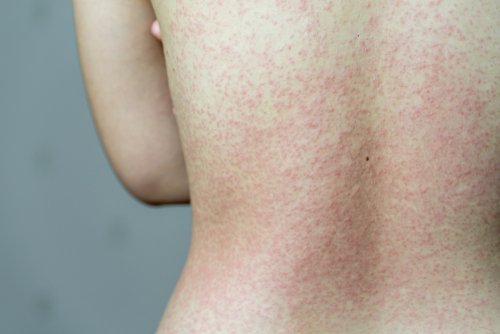
"The benefit of molnupiravir may be more limited. "Nirmatrelvir-ritonavir seems to be an effective treatment for eligible persons with COVID-19 to reduce risk for short-term outcomes of severe COVID-19," the authors write. An early report from dermatologists working with COVID-19 patients in Italy found that, in a group of 88 confirmed positive patients, 20 developed skin symptoms, with a little under half developing a rash at the onset of disease, and a little more than half developing it after hospitalization. No difference was seen in 30-day or 31- to 180-day risk for hospitalization or death between nirmatrelvir- or molnupiravir-treated participants. People who develop a skin rash a few weeks after clearing the COVID-19 infection might need immediate medical attention for a condition called MIS-A. Most people with COVID-19 experience fever, fatigue, and respiratory symptoms. Lower 30-day and 31- to 180-day risks for deaths were seen for molnupiravir-treated participants (3.14 versus 13.56 per 1,000 participants at 30 days hazard ratio, 0.67 at 31 to 180 days), but not for hospitalization. COVID-19 is a respiratory infection caused by the SARS-COV-2 virus. Reductions in 31- to 180-day incidence of death were seen among persons alive at day 31 (hazard ratio, 0.66), but not in hospitalization. 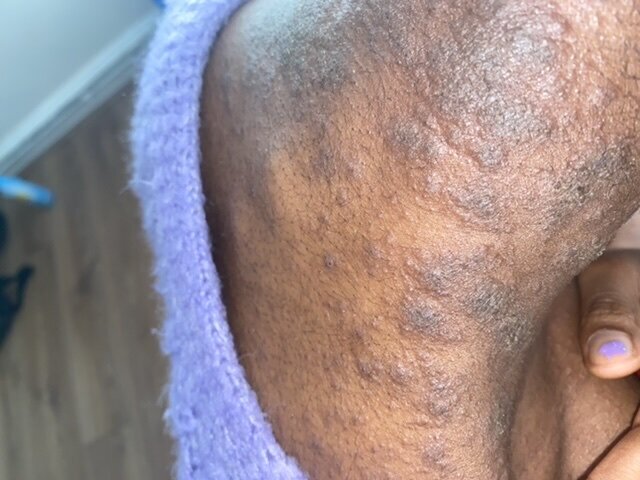
The researchers found that the 9,607 patients treated with nirmatrelvir-ritonavir had lower 30-day risk for hospitalization compared with matched untreated controls (22.07 versus 30.32 per 1,000 participants) and a lower risk for death (1.25 versus 5.47 per 1,000 participants). You have had any type of immediate (within 4 hours) allergic reaction to a non-COVID-19 vaccine or injectable therapy. You should be monitored for 30 minutes if: You have had a severe allergic reaction called anaphylaxis due to any cause. Overall, 87 percent of the participants were male, with a median age of 66 years 18 percent were unvaccinated. Everyone who gets a COVID-19 vaccine should be monitored on site for at least 15 minutes after vaccination. Bajema, M.D., from Oregon Health & Science University in Portland, and colleagues examined the effectiveness of nirmatrelvir-ritonavir and molnupiravir for outpatient treatment of COVID-19 in three retrospective target trial emulation studies involving nonhospitalized veterans at risk for severe COVID-19 who tested positive for severe acute respiratory syndrome coronavirus 2. For adults with COVID-19, symptoms may be more severe and may include trouble breathing. In children, COVID-19 often results in mild symptoms such as fever, runny nose and cough. Feeling sick or overly exhausted after physical activity, also known as post-exertional malaise.Kristina L. Because RSV and coronavirus disease 2019 (COVID-19) are both types of respiratory viruses, some symptoms of RSV and COVID-19 can be similar.In adults and older, healthy children, RSV symptoms are mild and typically mimic the common cold. Respiratory syncytial (sin-SISH-ul) virus can also infect adults. 
Its so common that most children have been infected with the virus by age 2. While hundreds of symptoms possibly associated with long Covid have long been reported - mostly through anecdotes from patients - the new study homes in on 12 of the most common. Overview Respiratory syncytial virus (RSV) causes infections of the lungs and respiratory tract. The vast majority, 8,646 people, had previously been diagnosed with Covid. The new study, published Thursday in the Journal of the American Medical Association, looked at data from 9,764 adults in the RECOVER trial, which has been recruiting participants since last year. The publics heightened attention to identifying the causes of infections, including the COVID-19 pandemic, has led to a greater focus on other viruses and the diseases they cause. 
Leora Horwitz, a professor of population health and medicine at New York University Grossman School of Medicine, as well as the director of the Center for Healthcare Innovation and Delivery Science at NYU Langone Health. A booster dose of Spikevax may be given to adults and children from the age of 6 years, at least 3 months after primary vaccination with Spikevax, or another. "I think that the amount of science that's going to come after this is going to be an avalanche," said study author Dr.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
